 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
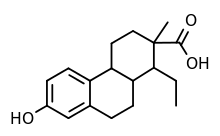
| Formula | C18H24O3 |
| Molar mass | 288.387 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Doisynolic acid is a synthetic, nonsteroidal, orally active estrogen that was never marketed.[1][2][3] The reaction of estradiol or estrone with potassium hydroxide, a strong base, results in doisynolic acid as a degradation product, which retains high estrogenic activity, and this reaction was how the drug was discovered, in the late 1930s.[4][5][6] The drug is a highly active and potent estrogen by the oral or subcutaneous route.[4] The reaction of equilenin or dihydroequilenin with potassium hydroxide was also found to produce bisdehydrodoisynolic acid, the levorotatory isomer[7] of which is an estrogen with an "astonishingly" high degree of potency, while the dextrorotatory isomer is inactive.[4] Doisynolic acid was named after Edward Adelbert Doisy, a pioneer in the field of estrogen research and one of the discoverers of estrone.[8]
Doisynolic acid is the parent compound of a group of synthetic, nonsteroidal estrogens with high oral activity.[9] The synthetic, nonsteroidal estrogens methallenestril, fenestrel, and carbestrol were all derived from doisynolic acid and are seco-analogues of the compound.[10] Doisynoestrol, also known as fenocycline, is cis-bisdehydrdoisynolic acid methyl ether, and is another estrogenic derivative.[11]
See also
References
- ↑ Hill RA, Makin HL, Kirk DN, Murphy GM (23 May 1991). Dictionary of Steroids. CRC Press. pp. 422–. ISBN 978-0-412-27060-4.
- ↑ Leclerc G (2 December 2012). "Estrogens, Antiestrogens, and Other Estrane Compounds". In Blickenstaff RT (ed.). Antitumor Steroids. Academic Press. pp. 11–12. ISBN 978-0-323-13916-8.
- ↑ Hilf R, Wittliff JL (27 November 2013). "Mechanims of Action of Estrogens". In Sartorelli AC, Johns DG (eds.). Antineoplastic and Immunosuppressive Agents. Springer Science & Business Media. pp. 106–. ISBN 978-3-642-65806-8.
- 1 2 3 Pearlman WH (2 December 2012). "The Chemistry and Metabolism of the Estrogens". In Pincus G, Thimann KV (eds.). The Hormones V1: Physiology, Chemistry and Applications. Elsevier. pp. 364–366. ISBN 978-0-323-14206-9.
- ↑ Blickenstaff RT, Ghosh AC, Wolf GC (22 October 2013). "Chapter 3.4: 18,19-Norprogesterone and 19-Norpreganes". Total Synthesis of Steroids: Organic Chemistry: A Series of Monographs. Elsevier Science. pp. 65–. ISBN 978-1-4832-1642-3.
- ↑ Brueggemeir RW, Miller DD, Dalton JT (January 2002). "Estrogen, Progestins, and Androgens". In Williams DA, Foye WO, Lemke TL (eds.). Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins. pp. 692–. ISBN 978-0-683-30737-5.
- ↑ "Doisynolic acid". Miraculous Medicines NFT. #3772. Retrieved 2023-11-02.
- ↑ Dodds C (May 1955). "Synthetic oestrogens". British Medical Bulletin. 11 (2): 131–134. doi:10.1093/oxfordjournals.bmb.a069465. PMID 14378564.
- ↑ Frieden E (2 December 2012). "Estrogens". Chemical Endocrinology. Elsevier Science. pp. 53–. ISBN 978-0-323-15906-7.
- ↑ Kirk RE, Othmer DF (1980). Encyclopedia of chemical technology. Wiley. p. 670,672. ISBN 978-0-471-02065-3.
- ↑ Elks J (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 465–. ISBN 978-1-4757-2085-3.